 
For example, one of the best known compounds is water, written chemically as H 20, which means that water is made of two atoms of hydrogen chemically bonded to one of oxygen. These are: antimony, arsenic, carbon, copper, iron, gold, lead, mercury, silver, sulfur, and tin.Īll other elements have been discovered since 1669: this was the year Hennig Brand became the first known person to discover a new element - phosphorus.Īn element can combine with one or more other elements to form compounds, of which there are millions. Some elements have been known for thousands of years, and we do not know who discovered them. We use the periodic table to display all of the elements in an organized way. There are currently 118 accepted elements. 
For example, element 1 is hydrogen, element 2 is helium, element 3 is lithium, element 8 is oxygen, etc. We can also name elements using their atomic numbers. Some of these names are familiar to us, such as nitrogen and sodium, and some are less familiar, such as dysprosium and roentgenium. 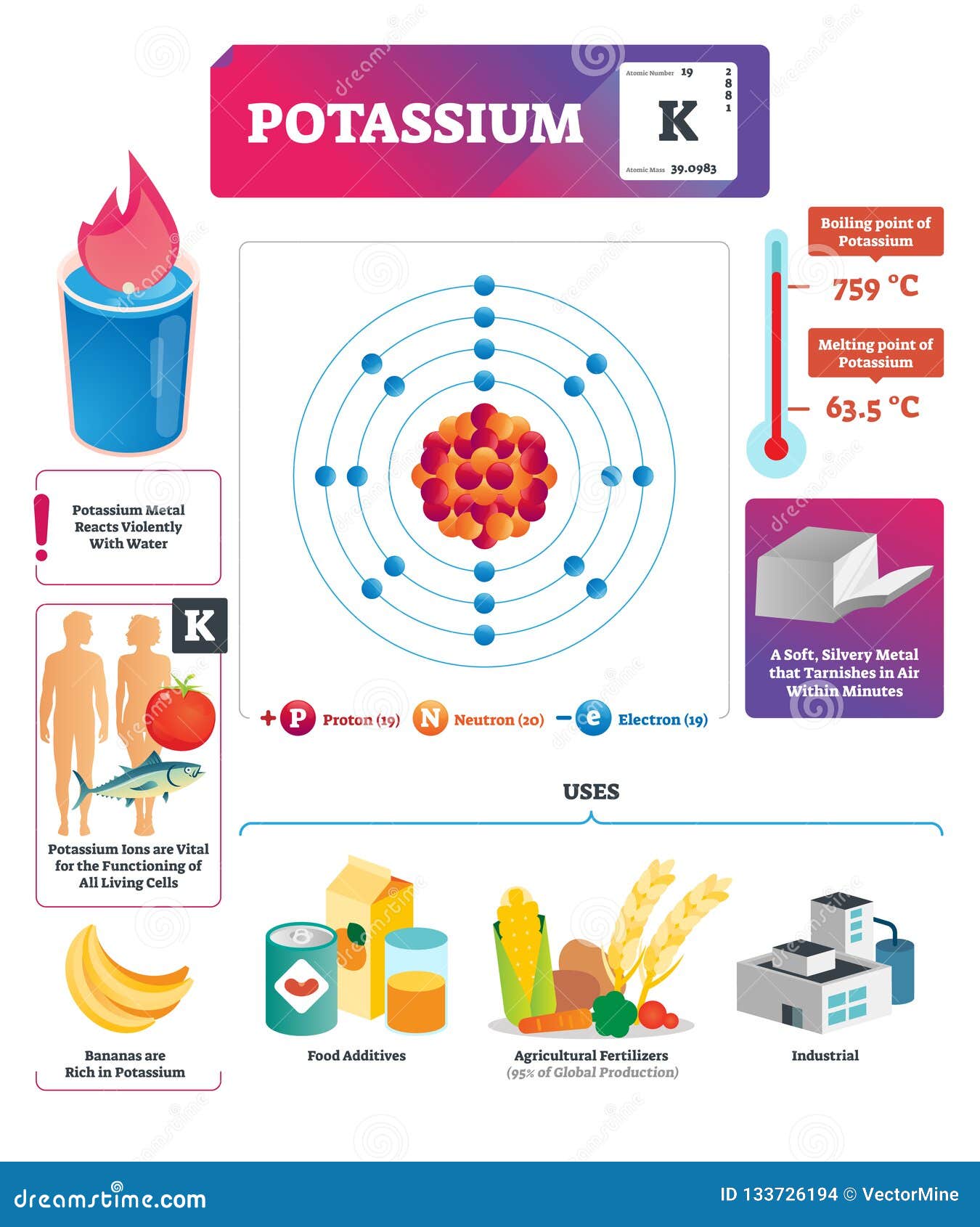
In the earth’s crust, oxygen (47%) is the most abundant element, followed by silicon (28%) and aluminum (8%).Īll of the elements have been named. Elements Appearance API Customize the look and feel of Elements to match the design of your site. To share feedback with us, Contact the Microsoft Edge DevTools team. Providing feedback about the experiments Were eager to hear your feedback about experimental features. To end filtering, clear the Filter text box. All of the other elements make up the remaining 1 percent. As you type, only the matching checkboxes are shown in the Experiments page. The crystal structure of Actinium is FCC (i.e face centered cubic). The atomic mass of the most stable isotope of actinium is 227 u and its density is 10.07 g/cm 3. In a dark room, actinium glows blue in color. Oxygen atoms have eight protons.Īt 75 percent, hydrogen is the most abundant element in the universe, followed by helium at 23 percent, then oxygen at 1 percent. Actinium is a radioactive element having a silvery white appearance. With only one proton, hydrogen is the simplest, lightest element, followed by helium, which has two protons. This is the story of how Henry Moseley brought light to the darkness. Visualizing the atom was a free-for-all, and Mendeleev's justification for a periodic table based on the elements' atomic weights was falling apart at the seams. Some big hitters - including Dmitri Mendeleev - were talking seriously about elements lighter than hydrogen and elements between hydrogen and helium. Published on Thursday, Updated on Thursday, September 19, 2019. In 1913, chemistry and physics were topsy-turvy. How Elements Came to be Defined Correctly When atoms of the same element have different numbers of neutrons, they are called isotopes. Elements can only be changed into other elements using nuclear methods.Īlthough an element’s atoms must all have the same number of protons, they can have different numbers of neutrons and hence different masses. An element is a substance whose atoms all have the same number of protons: another way of saying this is that all of a particular element's atoms have the same atomic number.Įlements are chemically the simplest substances and hence cannot be broken down using chemical reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
